sshaynijaonnahayde
13.12.2019 •
Chemistry
When thinking about the amount of solute present in a solution, chemists report the concentration or molarity of the solution. molarity is calculated as moles of solute per liter of solution. what is the molarity of the solution described above?
Solved
Show answers
More tips
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
- A Art and Culture When Will Eurovision 2011 Take Place?...
- S Style and Beauty How to Choose the Perfect Hair Straightener?...
- F Family and Home Why Having Pets at Home is Good for Your Health...
- H Health and Medicine How to perform artificial respiration?...
Answers on questions: Chemistry
- C Chemistry 2. I have a mixture of gasses that is made up of 2.3 moles of oxygen, 4.1 moles of bromine, 1.6 moles of iodine, and 0.7 moles of nitrogen, what is the mole fraction of each gas?...
- C Chemistry Finish the graph given ASAP please?...
- C Chemistry 3. I have 10 moles of a gas mixture. If there are 2.7 moles of hydrogen gas in the mixture and the gas is exerting a pressure of 115,000 pascals, what is the partial pressure of...
- C Chemistry How many neutrons are present in the atom of a team that has the atomic number of 50 and the mass number of 119?...
- C Chemistry Which of these is a property of a substance that is composed of atoms that are held together by metallic bonds? it has a low melting point. it can be flattened and bent. it cannot...
- C Chemistry An opened bottle of milk in a refrigerator stays fresh longer than one at room temperature on a table Assuming that losing freshness is related to a chemical reaction, which statement...
- C Chemistry Which procedure would produce the best data for comparing the electrostatic forces between the molecules of different substances? Heat each substance in liquid form for the same...
- C Chemistry Which phrase best describes the vapor point of a substance? the strength of the bonds between molecules of a substance that is in a gaseous state the amount of heat energy required...
- C Chemistry A total of 6.28 x 10^5 J is taken from a heat source at 150.0 degrees Celsius and rejected to a heat sink at 50.0 degrees Celsius. Find (a) the change of entropy of in the heat...
- C Chemistry An gel ice pack getting cold after you put it in the freezer for an hour. substance change chemical change elemental change physical change...

Ответ:
Completed question:
When chemists work with solid materials, we simply weigh out amounts of solid reagents and calculate mole amounts when thinking about stoichiometry. However, when we dissolve a solid (also known as a solute) in a solvent to form a solution, the solute becomes evenly distributed throughout the solution and we need to know how many moles of solute are present in a particular volume of solution. A solution is created by dissolving 11.0 grams of ammonium chloride in enough water to make 215 mL of solution.
How many moles of ammonium chloride are present in the resulting solution? ___moles of NH4Cl
When thinking about the amount of solute present in a solution, chemists report the concentration or molarity of the solution. Molarity is calculated as moles of solute per liter of solution. What is the molarity of the solution described above? ____M
Number of moles of NH4Cl = 0.2056moles
Molarity = 0.956M
Explanation:
Number of moles =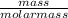
molar mass of NH4Cl = 14 +(4*1) +35.5 = 53.5g/mol
mass of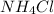 = 11g
= 11g
number of moles of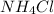 =
= 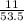
= 0.2056 moles
Molarity of a solution refers to the number of moles present in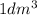 of solution.
of solution.
if 0.2056 moles is present in 215mL of solution, we will find how many moles will be present in 1L of solution
=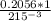
= 0.956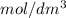
= 0.956M solution.
Ответ:
Explanation:
Temperature