Which of the following includes an example of a chemical property of an element
a.
Solved
Show answers
More tips
- H Horoscopes, Magic, Divination Where Did Tarot Cards Come From?...
- S Style and Beauty How to Make Your Lips Fuller? Ideas and Tips for Beautiful Lips...
- S Style and Beauty How are artificial nails removed?...
- F Family and Home How to Sew Curtain Tapes: Best Tips from Professionals...
- H Horoscopes, Magic, Divination How to Cast a Love Spell on a Guy? Guide for Guys...
- F Family and Home How to Properly Use a Water Level?...
- L Legal consultation What Documents Are Required for a Russian Passport?...
- H Health and Medicine How to Treat Styes: Causes, Symptoms, and Home Remedies...
- F Family and Home Protect Your Home or Apartment from Pesky Ants...
- T Travel and tourism Lost in the Catacombs: What to Do?...
Answers on questions: Chemistry
- M Mathematics What is true about the dilation?...
- H Health True or false some of the physiological effects of cortisol and adrenaline are; increased heart rate and energy and a quicker reaction time....
- E English Read the excerpt from a short story...
- M Mathematics What is 10.8 as a mixed number...

Ответ:
It includes flammability ,toxicity,acidity reactivity..And heats of combustion.Iron ..
If this answer helps you plz mark as brainlist..
tq
Ответ:
Solution :
For the reaction given :
Thus we know that the equilibrium constant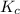 contains aqueous an dgas species only.
contains aqueous an dgas species only.
∴![$K_c=\frac{1}{[O_2]}$](/tpl/images/1168/7990/eab15.png) ............(1)
............(1)
Now at the equilibrium, an amount of the 13.4 g of oxygen was found in the vessel of 6.9 liters. For determining the concentration of the oxygen gas, we use :
Here,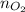 = no. of moles of oxygen gas (mol)
= no. of moles of oxygen gas (mol)
Therefore the number of moles of the oxygen gas is calculated by directly using the molecular weight (31.9988 g/mol) as the conversion factor.
∴
= 0.418 mol
Now substituting the known values in (2), we can find the equilibrium concentration of the oxygen gas :
= 0.0605 M
Therefore substituting the result in (1), the equilibrium constant for the reaction is :
= 16.52