kannstyles709
22.06.2021 •
Chemistry
You performed fat analysis on a new super energy shake (high carbohydrate and protein)
using standard Soxhlet extraction. The value obtained for fat content was much lower than
that expected. What could have caused the measured fat content to be low, and how would
you modify the standard procedure to correct the problem?
Solved
Show answers
More tips
- G Goods and services Should You Engrave Your Wedding Rings? Facts, Myths, and Considerations...
- L Leisure and Entertainment How to Land on the Moon: Your Comprehensive Guide...
- T Travel and tourism How to Use a Compass: A Beginner s Guide...
- C Computers and Internet Porn Banner: What It Is and How to Get Rid Of It?...
- C Computers and Internet Отправляем смс через интернет: легко и просто...
- L Leisure and Entertainment The Best Film of 2010: A Look Back at the Academy Awards...
- H Health and Medicine Simple and Effective: How to Get Rid of Cracked Heels...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry In a parallel circuit, what happens to the overall current of the circuit if the number of receivers (and thus the number of branches) is doubled?...
- C Chemistry T6hw question 22 - challenge homework • unanswered the enthalpy of atomization is the enthalpy required to convert a compound into gaseous monatomic atoms by breaking...
- C Chemistry Which of the following steps is usually done right before experimentation in the scientific method of investigation? construct a hypothesisask a questiondo background...
- C Chemistry Give example of an insoluble base by the action of a soluble base on a soluble salt...
- C Chemistry Acombustion reaction involves the reaction of a substance with oxygen gas. the complete combustion of any hydrocarbon (binary compound of carbon and hydrogen) produces...
- C Chemistry A saturated solution of NaNo3 in 50 grams of water at 10.0C is heated to 40.0C. Approximately how much more NaNO3 must be added to keep the solution saturated at the...
- C Chemistry Which of the statements about peptide bonds are true? The formation of a peptide bond involves a dehydration reaction. A tripeptide contains three amino acid residues....
- C Chemistry After 11.5 days, 12.5% of a sample of radon-222 that originally weighed 42g remains. what is the half-life of this isotope?...
- C Chemistry Poem about how ones lifestyle can affect the circulatory and respiratory system...
- M Mathematics In a triangle, the measure of the first angle is three times the measure of the second angle. The measure of the third angle is 80 degrees more than the measure of...

Ответ:
gdhdj7shwmaogsbsjdyjwjs
Ответ:
This question is incomplete, the complete question is;
The times of first sprinkler activation (in seconds) for a series of tests of fire-prevention sprinkler systems that use aqueous film-forming foam is as follows; 27 41 22 27 23 35 30 33 24 27 28 22 24
( see " use of AFFF in sprinkler systems," Fire technology, 1975: 5)
The system has been designed so that the true average activation time is supposed to be at most 25 seconds.
Does the data indicate the design specifications have not been met?
Test the relevant hypothesis at significance level 0.05 using the P-value approach
since p-value (0.042299) is lesser than the level of significance ( 0.05)
We Reject Null Hypothesis
Hence, There is no sufficient evidence that the true average activation time is at most 25 seconds
Explanation:
Given the data in the question;
lets consider Null and Alternative hypothesis;
Null hypothesis H₀ : There is sufficient evidence that the true average activation time is at most 25 seconds
Alternative hypothesis H₁ : There is no sufficient evidence that the true average activation time is at most 25 seconds
i.e
Null hypothesis H₀ : μ ≤ 25
Alternative hypothesis H₁ : μ > 25
level of significance σ = 0.05
first we determine the sample mean;
where n is sample size and ∑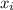 is summation of all the sample;
is summation of all the sample;
=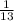 ( 27 + 41 + 22 + 27 + 23 + 35 + 30 + 33 + 24 + 27 + 28 + 22 + 24 )
( 27 + 41 + 22 + 27 + 23 + 35 + 30 + 33 + 24 + 27 + 28 + 22 + 24 )
=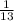 ( 363
( 363
sample mean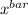 = 27.9231
= 27.9231
next we find the standard deviation
s = √(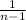 ∑(
∑(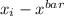 )²
)²
x (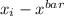 ) (
) (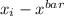 )²
)²
27 -0.9231 0.8521
41 13.0769 171.0053
22 -5.9231 35.0831
27 -0.9231 0.8521
23 -4.9231 24.2369
35 7.0769 50.0825
30 2.0769 4.3135
33 5.0769 25.7749
24 -3.9231 15.3907
27 -0.9231 0.8521
28 0.0769 0.0059
22 -5.9231 35.0831
24 -3.9231 15.3907
sum 378.9229
so ∑(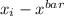 )² = 378.9229
)² = 378.9229
∴
s = √(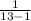 ×378.9229 )
×378.9229 )
s = √31.5769
standard deviation s = 5.6193
now, the Test statistics
t = (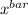 - μ ) /
- μ ) / 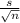
we substitute
t = ( 27.9231 - 25 ) /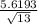
t = 2.9231 / 1.5585
t = 1.88
now degree of freedom df = n - 1 = 13 - 1 = 12
next we calculate p-value
p-value = 0.042299 ( using Execl's ( = TDIST(1.88,12,1)))
Here x=1.88, df=12, one tail
now we compare the p-value with the level of significance
since p-value (0.042299) is lesser than the level of significance ( 0.05)
We Reject Null Hypothesis
Hence, There is no sufficient evidence that the true average activation time is at most 25 seconds