travisalier4171
02.12.2020 •
Chemistry
A bottle of hydrogen peroxide (H2O2) is labeled 3.0% (v/v). How many mL H2O2 are in a 400.0-ml bottle?
Solved
Show answers
More tips
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- S Social Studies What role did slavery play in the American colonial economy?...
- M Mathematics 7. Select all statements that are true of the graph shown. A. The rate of the increase is greater than the rate of the decrease. B. The first segment of the graph...
- M Mathematics The diagonal of a rectangle is 40 and its base is 32. find the altitude of the rectangle....
- M Mathematics Would you rather be able to breath underwater or fly through the air?...
- H Health Nterviewee Name: Age: Gender: Job: Date and Time of interview: 1.What are some major health concerns that you have? 2.Are there ways that you know you can monitor...

Ответ:
La temperatura final del sistema es 1029,346 °C.
Explanation:
Asumamos que el sistema conformado por el cobre y el estaño no tiene interacciones con sus alrededores. Por la Primera Ley de la Termodinámica, el cobre cede calor al estaño con tal de alcanzar el equilibrio térmico. El cobre se encuentra inicialmente en su punto de fusión, mientras que el estaño está por encima de ese punto, de modo que la transferencia de calor es esencialmente sensible:
Donde:
Si sabemos que ,
,  ,
,  ,
, 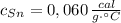 ,
, 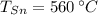 y
y  , entonces la temperatura final del sistema es:
, entonces la temperatura final del sistema es:
La temperatura final del sistema es 1029,346 °C.