According to the rate law, how do concentrations affect rate?
Solved
Show answers
More tips
- F Food and Cooking How to Make Shortcrust Pastry: Recipe and Tips...
- H Horoscopes, Magic, Divination Is there a 13th Zodiac Sign?...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry 11. If you have three moles of S., how many moles of Als, can be produced?...
- C Chemistry A rigid tank contains an ideal gas at 40°C that is being stirred by a paddle wheel. The paddle wheel does 150 kJ of work on the ideal gas. It is observed that the temperature...
- C Chemistry The pressure at the center of the earth is probably greater than 3x106 atm, and the temperature there is about 4000 °C. What is the change in the Gibbs function of reaction...
- C Chemistry If 1.881 M of KF has a volume of 296.8 mL. What mass of KF is in the solution....
- C Chemistry Economics At which point would the government be most likely to try to stimulate the economy?124 At which point would a business be most likely to hire new workers?...
- C Chemistry Use this information to calculate AGxn 2C_H2(g)+50 (9) — 4C02(g)+ 2H20(9) AG CHą = 209.2 kJ/mol AGE.CO. = -394.4 kJ/mol AG 4,0 = -228.57 kJ/mol TELE ebo.. AGx = kJ...
- C Chemistry Chlorine and hydrogen gas react to form hydrogen chloride as shown in the following reaction: Cl2 (g) + H2 (g) → 2HCI (g) If a 48.5 L sample of chlorine gas was reacted...
- C Chemistry How do plants use the stored sugars that are formed in photosynthesis?...
- C Chemistry Can someone help me on this pls?asap pls...
- C Chemistry a flask of water is sealed with a balloon and heated to various tempatures on a hot plate. Fill in the missing info below...

Ответ:
Explanation:
According to rate law, rate of a reaction depends upon the concentration of the species participating in the reaction.
For example,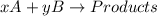
Rate =![k[A]^{x}[B]^{y}](/tpl/images/0085/9900/8fb52.png)
Therefore, when there is increase in concentration of reactants then there will be more number of collisions. Due to this there will rapid formation of products. Hence, rate of reaction will increase.
Whereas decrease in concentration of reactants will decrease the rate of a reaction.
Ответ:
All the elements of a group have similar chemical properties because they have same no. of valence electrons in their outermost shell.
All the elements of a period have different chemical properties because they have different no. of valence electrons in their atoms.