danielaguardado63
16.12.2020 •
Chemistry
Explain what the difference is between chemical properties, extensive physical properties, and intensive physical properties. ill award brainliest for right answer
Solved
Show answers
More tips
- S Style and Beauty How to Apply Foundation Correctly?...
- A Animals and plants How to Store Carrots: Tips for Homeowners...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
Answers on questions: Chemistry
- C Chemistry 1 Four substances were mixed with a clear liquid. The observations are recorded in the table shown below. Substance Appearance Before Mixing Appearance of Mixture Pale...
- E English 19 Which of the following is the best synonym for monotonous ? A. varied B. boring 1, C. exciting D. interesting...
- H History Please help me and if you are right and hurry I ll give you an extra 20 points! Activity Now it’s your turn to write about an event or idea from two different perspectives....
- E English What are the women s right included by un...

Ответ:
Extensive properties, such as mass and volume, depending on the amount of matter being measured. Intensive properties, such as density and color, do not depend on the amount of the substance present. Physical properties can be measured without changing a substance's chemical identity.
Explanation:
Ответ:
The enthalpy of the reaction for given amount of ammonia will be -3431.3 kJ.
Explanation:
To calculate the number of moles, we use the equation:
For ammonia:
Given mass of ammonia =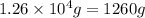
Molar mass of ammonia = 17 g/mol
Putting values in above equation, we get:
We are given:
Moles of ammonia = 74.11 moles
For the given chemical reaction:
By Stoichiometry of the reaction:
If 2 moles of ammonia produces -92.6 kJ of energy.
Then, 74.11 moles of ammonia will produce = of energy.
of energy.
Thus, the enthalpy of the reaction for given amount of ammonia will be -3431.3 kJ.