Half- fill a plastic cup with warm water Cover the cup with plastic wrap. Secure the plastic with a rubber band. Place an ice cube on the plastic wrap. Observe the cup for several minutes. Record your observation below
Solved
Show answers
More tips
- F Food and Cooking What s the Best Rice for Cooking Plov?...
- F Family and Home How to Remove Fading from Clothes: Tips and Tricks...
- F Food and Cooking How to Make Polendwitsa at Home?...
- F Family and Home Parents or Environment: Who Has the Most Influence on a Child s Upbringing?...
- P Philosophy Unbelievable stories of encounters with otherworldly forces...
- L Leisure and Entertainment How to Choose the Perfect Gift for Men on February 23rd?...
- H Health and Medicine How to Treat Whooping Cough in Children?...
- H Health and Medicine Simple Ways to Lower Cholesterol in the Blood: Tips and Tricks...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Calculate the molar solubility of silver chloride in 0.15 m sodium chloride. ksp of silver chloride = 1.6x10-10....
- C Chemistry {Blank} are used to rétard energy loss. A. ConductorsB. RadiationC. Insulators D. Currents...
- C Chemistry Cual es mejor fusión nuclear o fisión nuclear...
- C Chemistry IS A TOMATO A FRUIT OR VEGETABLE...
- C Chemistry During the rGFP purification experiment, the instructor will have to make breaking buffer for the students to use. This buffer contains 150mM NaCl. Given a bottle of crystalline...
- C Chemistry What is the empirical formula of a compound composed of 28.7 g of potassium ( K ) and 5.87 g of oxygen ( O )? Insert subscripts as needed....
- C Chemistry Chemistry. can someone pls check out my answers and make sure that they are all correct. i need a 100% on this quiz and it is due tonight. youuu...
- C Chemistry A certain chemical reaction releases of heat for each gram of reactant consumed. How can you calculate the heat produced by the consumption of of reactant? Set the math up....
- C Chemistry Please i really need help! :( Please help me this is due at 8:45 pleaseeeee...
- M Mathematics PLZ HEL Find the coordinates of the vertices of the triangle if it’s rotated 270° counterclockwise. A.X′(–1,3), Y′(–5,2), Z′(–3,–1) B.X′(3,–1), Y′(2,–5), Z′(–1,–3) C.X′(–3,1),...

Ответ:
Answer : The correct option is, (A) 82.35% N; 17.65% H
Explanation :
Percent composition : It is calculated by dividing the mass of each element in one mole of the compound that means dividing the mass of each element by the total molar mass of the compound.
Formula used :
The given molecule is,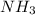
Molar mass of H = 1 g/mole
Molar mass of N = 14 g/mole
First we have to calculate the molar mass of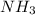 .
.
Molar mass of glucose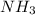 =
= 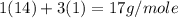
Now we have to calculate the percent composition of nitrogen and hydrogen in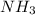 .
.
As we now that there are 1 number of nitrogen atom and 3 number of hydrogen atoms.
Now put all the given values in this formula, we get:
and,
Now put all the given values in this formula, we get:
Therefore, the percent composition of nitrogen and hydrogen in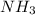 is, 82.35 % and 17.65 % respectively.
is, 82.35 % and 17.65 % respectively.