Jerryholloway5871
20.06.2020 •
Chemistry
How do you complete this equation
Solved
Show answers
More tips
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- E English The dewey decimal system is the classified system used by...
- C Computers and Technology All the shortys comment your snapcht below :o...
- G Geography Which if the following is not a principle group in the koppen system?...
- M Mathematics What is the decimal value of the fraction 2/7 ? a)0.285714¯ b)0.285714¯¯¯¯¯¯¯¯¯¯ c)0.285714 d)0.2857142857¯¯¯¯¯...

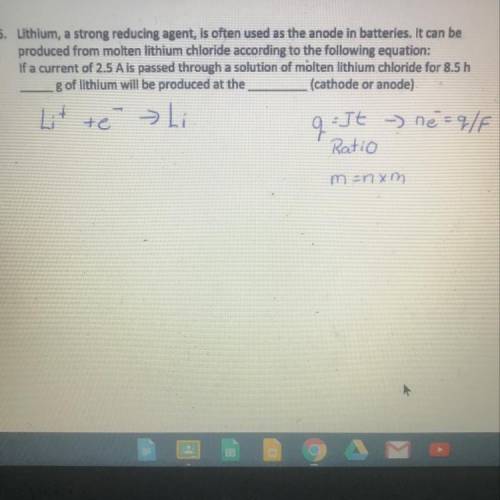
Ответ:
5.55g of Li will be deposited at the cathode.
Explanation:
Step 1:
Data obtained from the question. This include:
Current (I) = 2.5A
Time (t) = 8.5hrs = 8.5 x 60 x 60 = 30600 secs
Step 2:
Determination of the quantity of electricity.
The quantity of electricity, Q can be obtained as follow:
Q = it
Q = 2.5 x 30600
Q = 76500C
Step 3:
Determination of the number of faraday needed to deposit metallic lithium.
Lithium is a univalent metal and will be deposited in solution as follow:
Li+ e- —> Li
From the above, 1 faraday is required to deposit metallic lithium.
1 faraday = 96500C
Molar mass of Li = 7g/mol
From the balanced equation above,
96500C of electricity is required to deposit 7g of Li.
Step 4:
Determination of the mass of lithium deposited during the process. This is illustrated below:
From the balanced equation above,
96500C of electricity is required to deposit 7g of Li.
Therefore, 76500C of electricity will deposit = (76500 x 7)/96500 = 5.55g of Li.
Therefore, 5.55g of Li will be deposited at the cathode.
Note: the cathode is the negative electrode and Li being a metal will migrate to the cathode while chlorine being a non metal will migrate to the positive electrode(anode).
Ответ:
1.They break the bonds between the ions.
2.They remove electrons from the ions.
Explanation:
covalent bonds do not dissolve in water but some covalent compoound s do