lailahussain99
28.09.2019 •
Chemistry
How would you prepare 250.0 ml of 0.00200 m na2s2o3? describe what you would do in the lab. include amounts and types of glassware and equipment that you would use.
Solved
Show answers
More tips
- H Horoscopes, Magic, Divination Is there a 13th Zodiac Sign?...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry Un elemento sólido tiene un punto alto de fusión y por sus características es usado dentro de artefactos electrónicos. Probablemente es un...
- C Chemistry Sodium (Na) and iodine gas (I2) react to form sodium iodine (NaI). Which equation is a correct representation of the balanced equation? 2Na+I2→2NaI I2→2NaI+2Na Na→NaI+I2...
- C Chemistry What is electron affinity?...
- C Chemistry What is the relationship between wavelength and energy in electromagnetic waves?...
- C Chemistry Students set up an experiment to investigate whether or not a chemical reaction has occurred when mixing an antacid tablet with water. The results are listed in the table....
- C Chemistry - How many grams are in 1.40 moles of NO2? Please help quick...
- C Chemistry Ionic bonds are formed between atoms with A. similar haircuts B. a medium to large electronegative difference C. a zero to small electronegative difference D. a medium electronegative...
- C Chemistry Acompound analyzed as 18.0% carbon, 2.26% hydrogen, and 79.7% chlorine. calculate the empirical formula of the compound....
- M Mathematics PLEASE HELP QUICK WILL MARK BRANLIEST...
- M Mathematics A cake recipe says to bake the cake until the center is 180°F. then let the cake cool to 100°F. The table shows temperature readings for the cake. a. Given a room temperature...

Ответ:
You have to weigh 0.079 g of Na₂S₂0₃ and dissolve it in water at a final volume of 250 ml.
Explanation:
A 0.00200 M Na₂S₂O₃ solution has 0.002 mol of Na₂S₂O₃ per liter of solution. As we know that 1 L = 1000 ml, and that the molecular weight of Na₂S₂O₃ IS 158 g/mol, we can calculate the mass of Na₂S₂O₃ to weigh as follows:
mass=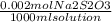 x
x 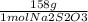 x 250 ml solution
x 250 ml solution
mass= 0.079 g
To prepare the solution, we have to weigh in a beaker with 0.079 g of Na₂S₂O₃ by employing an analytical balance. Then, we have to dissolve the mass in a volume of aproximately 230 ml water. For this, we measure the 230 ml of water in a graduated cylinder, we add the volume to the beaker with the mass and we agitate until total disolution. Finally, we tranfer the total amount of the solution in the glass to a volumetric flask with a capacity of 250 ml. We add water until we reach the capacity, and then we homogenize the solution.
Ответ: