ayoismeisjjjjuan
06.01.2022 •
Chemistry
PLEASE HELP ASAP WILL GIVE BRAINLIEST
(dont give chegg link pls)
How much heat is required to convert 150 g of liquid H2O at 43.5 °C into steam at 130 °C?
Specific heat of liquid water is 4.18 J/g oC.
Specific heat of water vapor is 1.84 J/g oC.
ΔHvap = 40.67 kJ/mol
Solved
Show answers
More tips
- C Computers and Internet Е-head: How it Simplifies Life for Users?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry (20 points) In the morning, you decide to make toasted bacon, lettuce, and tomato sandwich. Observe what happens in each stage of making the sandwich. Which parts involve...
- C Chemistry A chemist conducts an experiment in which 2. 00 L of hydrogen gas is collected over water at 1. 00 atm and 298. 15 K. The phrase over water means that the gas was collected...
- C Chemistry An element is defined as a substance that is composed of only one type of atom. true or false?...
- C Chemistry Copper is an example of? Transition Metal Noble Gases Alkali Metal Halogen...
- C Chemistry When a 500. gram sample of water at 19.0°C absorbs 8400 Joules of heat, the temperature of the water will change to...
- C Chemistry 1. Explain taxonomy 2. How do we use taxonomy in everyday life...
- C Chemistry Which one is oxidised a which one is reduced...
- C Chemistry If a glacier is in equilibrium, where ablation is equal to the accumulation, what occurs at the terminus? A). the glacier is growing larger B). the glacier stops moving...
- C Chemistry Question 1: Would dissolving sugar into a cup of coffee be considered a physical or chemical change? Explain. Question 2: Identify 3 physical changes that could occur with...
- C Chemistry How many moles of AlCl3 are in 2.25L of a 0.15 molar solution? A. 0.07 moles B. 0.15 moles C. 0.34 moles D. 15...

Ответ:
The amount of heat required to convert H₂O to steam is : 382.62 kJ
Given data :
Mass of liquid water ( m ) = 150 g
Temperature of liquid water = 43.5°C
Temperature of steam = 130°C
Determine the amount of heat requiredThe amount of heat required = ∑ q1 + q2 + q3 ----- ( 1 )
where ;
q1 = heat required to change Temperature of water from 43.5°C to 100°C . q2 = heat required to change liquid water at 100°C to steam at 100°C
q3 = heat required to change temperature of steam at 100°C to 130°C
For q1M* S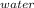 *ΔT
*ΔT
= 150 * 4.18 * ( 100 - 43.5 )
= 35425.5 J
For q2moles * ΔHvap
= (150 / 18 )* 40.67 * 1000
= 338916.67 J
For q3M * S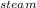 * ΔT
* ΔT
= 150 * 1.84 * ( 130 -100 )
= 8280 J
Back to equation ( 1 )
Amount of heat required = 35425.5 + 338916.67 + 8280 = 382622.17 J
≈ 382.62 kJ
Hence we can conclude that The amount of heat required to convert H₂O to steam is : 382.62 kJ.
Learn more about Specific heat of water :
Ответ:
Sorry don’t know
Explanation: