TURBONTRO6830
16.03.2020 •
Chemistry
PLEASE HELP FAST IM ABOUT TO FAIL
How many liters of water would be needed to dissolve 21.6 g of lithium nitrate to make a 1.3 M (molar) solution?
What is the molarity of a solution made of 215.1 g of HCl is dissolved to make 2.0 L of solution?
How much concentrated 18 M HSO is needed to prepare 250.0 mL of a 6.0 M solution?
A chemist has a stock solution of HBr that is 10.0 M and would like to make 450.0 mL of 3.0 M HBr, how would he/she do it?
How much water should be added to 50.0 mL of 12 M hydrochloric acid to make a 4.0 M solution?
Lulu Labwrecker carefully pipeta 25.0 mL of 0.525 M NaOH into a test tube. She places the test tube into a small beaker to keep it from spilling and then pipets 75.0 mL of 0.355 M HCl into another test tube. When Lulu reaches out to put this test tube of acid into the beaker along with test tube of base she accidentally knocks the test tubes together hard enough to break them and their respective contents combine in the bottom of the beaker. Is the solution formed from the contents of the two test tubes acidic or basic? What is the pH of the resulting solution?
Solved
Show answers
More tips
- F Food and Cooking How to Make Shortcrust Pastry: Recipe and Tips...
- H Horoscopes, Magic, Divination Is there a 13th Zodiac Sign?...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- M Mathematics What is the original quadratic equation of the image?...
- C Chemistry Which distance measures 5 units? the distance between points w and x the distance between points x and y the distance between points x and z the distance between points...
- M Mathematics The scatter plot shows the number of pumpkins that have been picked on the farm during the month of October: A scatter plot with points showing an upward trend with points...

Ответ:
1. 0.240 liters of water would be needed to dissolve 21.6 g of lithium nitrate to make a 1.3 M (molar) solution.
2. 2.9 M is the molarity of a solution made of 215.1 g of HCl is dissolved to make 2.0 L of solution.
3.83.3 ml of concentrated 18 M H2SO4 is needed to prepare 250.0 mL of a 6.0 M solution.
4. 135 ml of stock HBr will be required to dilute the solution.
5. 150 ml of water should be added to 50.0 mL of 12 M hydrochloric acid to make a 4.0 M solution
6. The pH of the resulting solution is 13.89
Explanation:
The formula used in solving the problems is
number of moles= 1st equation
1st equation
molarity =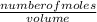 2nd equation
2nd equation
Dilution formula
M1V1 = M2V2 3rd equation
1. Data given
mass of Lithium nitrate = 21.6 grams
atomic mass of on emole lithium nitrate = 68.946 gram/mole
Molarity is given as 1.3 M
VOLUME=?
Calculate the number of moles using equation 1
n =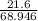
= 0.313 moles of lithium nitrate.
volume is calculated by applying equation 2.
volume =
= 0.240 litres of water will be used.
2. Data given:
mass of HCl = 215.1 gram
atomic mass of HCl = 36.46 gram/mole
volume = 2 litres
molarity = ?
using equation 1 number of moles calculated
number of moles =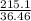
number of moles of HCl = 5.899 moles
molarity is calculated by using equation 2
M =
= 2.9 M is the molarity of the solution of 2 litre HCl.
3. data given:
molarity of H2SO4 = 18 M
Solution to be made 250 ml of 6 M
USING EQUATION 3
18 x V1= 250 x 6
V1 = 83.3 ml of concentrated 18 M H2SO4 will be required.
4. data given:
M1= 10M, V1 =?, M2= 3 ,V2= 450 ml
applying the equation 3
10 x VI = 3x 450
V1 = 135 ml of stock HBr will be required.
5. Data given:
V1 = 50 ml
M1= 12 M
V2=?
M2= 4
applying the equation 3
50 x 12 = 4 x v2
V2 = 150 ml.
6. data given:
HCl + NaOH ⇒ NaCl + H20
molarity of NaOH = 0.525 M
volume of NaOH = 25 ml
molarity of acid HCl= 75 ml
volume of HCl = 0.335 ml
pH=?
Number of moles of NaOH and HCl is calculated by using equation 1 and converting volume in litres
moles of NaOH = 0.0131
moles of HCl= 0.025 moles
The ratio of moles is 1:1 . To find the unreacted moles of acid and base which does not participated in neutralization so the difference of number of moles of acid minus number of moles of base is taken.
difference of moles = 0.0119 moles ( NaOH moles is more)
Molarity can be calculated by using equation 1 in (25 +75 ml) litre of solution
molarity =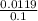
= 0.11 M (pOH Concentration)
14 = pH + pOH
pH = 14 - 0.11
pH = 13.89
Ответ:
to deal a blow or stroke to (a person or thing), as with the fist, a weapon, or a hammer; hit. to inflict, deliver, or deal (a blow, stroke, attack, etc.). to drive so as to cause impact: to strike the hands together. to thrust forcibly: Brutus struck a dagger into the dying Caesa
Explanation: