The rotten smell of a decaying animal carcass is partially due to a nitrogen-containing compound called putrescine. Elemental analysis of putrescine indicates that it consists of:
C
,
54.50
%
,
H
,
13.73
%
, and
N
,
31.77
%
. Calculate the empirical formula of putrescine.
Solved
Show answers
More tips
- H Horoscopes, Magic, Divination Is there a 13th Zodiac Sign?...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: Chemistry
- C Chemistry If you are needing at least 100. grams of HCI to be produced, how many grams of SiCl4 is t needed? * SiCl4(l) + _ H2O(l) —— SiO2 (s) + HCl(aq)...
- C Chemistry In which reaction is a precipitation occurring...
- C Chemistry Pure sugar is an example of a(n): element mixture solution compound...
- C Chemistry What happens to bases when mixed with acids?...
- C Chemistry If the fugacity coefficient of the components of a binary mixture are = 0.784, and =0.638 and mole fraction of component 1 is 0.4, write down and expression for the fugacity...
- M Mathematics $43 dinner; 18% gratuity whats the answer...
- M Mathematics An item is regularly priced at $40 it is on sale for %15 of the regular price. what is the sale price?...
- C Chemistry If a mixture of total pressure 50 psia obeys raoult s law and a species has a vapour presse of 20psia, what is the depriester k-value for the species in the mixture?...
- H Health He grieving process is a. the same for all people b. more effective when you have a strong support system c. more effective when you don t think about the loss d. only important...
- C Chemistry For an ideal gas mixture of a and b, if the total pressure is 100 kpa and component b has a mole fraction of 0.25, calculate the partial pressure of component...

Ответ:
The empirical formula is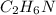 .
.
Explanation:
Putrescine has the elements like Carbon, Nitrogen and Hydrogen present in them. So in order to determine the empirical formula, we first have to find the number of moles present in the putrescine. As the percentage of C, H and N present in the chemical is given as 54.50%, 13.73% and 31.77%, we assume that 100 g of Putrescine is taken as sample.
Then the mass of C, H and N present in Putrescine will be 54.50 g, 13.73 g and 31.77 g. We know that the molar mass of C is 12 g/mol, H is 1 g/mol and N is 14 g/mol. So divide the mass with the molar mass of the respective elements to determine the number of moles of these elements present in the sample.
Similarly, the number of moles of H and N present is determined.
Then the empirical formula can be determined by dividing the number of moles of all elements with the least number of moles that is 2.27.
So, the empirical formula is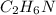 .
.
Ответ:
Carbon -14 and Carbon 12 are the two substances geologists use in radiocarbon dating.
Option B
Explanation:
Radiocarbon dating denotes the determination process of the age of fossils of plants or animals based on the ratio of carbon atoms 14 to 12. Carbon naturally exists in two non-radioactive isotopes, Carbon-12 and Carbon-13 and one radioactive isotope carbon 14. The carbon 14 gets released on continuous cosmic reaction with atmospheric nitrogen.
These carbon 14 will be absorbed by the living plants and from the plants. Then, it will enter inside the animals which consume the plants. But once the plants and animals died, they ceased to intake carbon-14. In their living state, the ratios of carbon atoms 14 to 12 in them tends to similar to the ratio in atmosphere.
But after they die, the ratio of C-14 to C-12 will be varying from the ratio of C-14 to C-12 in atmosphere as the concentration of C-14 will be decreasing in the dead animals and plants. Thus using this ratio, geologists can find the fossil's age.