clairajogriggsk
26.08.2019 •
Chemistry
What is the main result of respiration?
Solved
Show answers
More tips
- A Animals and plants How to Store Carrots: Tips for Homeowners...
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
Answers on questions: Chemistry
- C Chemistry Which statement best describes plastics...
- M Mathematics Formulate the situation as a system of two linear equations in two variables. Be sure to state clearly the meaning of your x- and y-variables. Solve the system by the elimination method....
- A Advanced Placement (AP) What is one reason that John Adams and Benjamin Franklin believed the Articles of Confederation should be revised? A. to establish the Constitutional Congress and its delegation B....
- E English Henry david thoreau: walden ch 2 excerpt the nation itself, with all its so-called internal improvements, which, by the way are all external and superficial, is just such an unwieldy...
- C Chemistry Radium decays to form radon. Which equation correctly describes this decay?...

Ответ:
6 O2 + C6H12O6 → 6 CO2 + 6H2O
Ответ:
Answer : For, this question,
The balanced equation is; 2Al + 3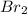 ----> 2Al
----> 2Al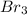
We can now, calculate how many grams of will be needed to completely convert 15 g of Al into Al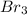 .
.
Here, the reaction produces, 2 moles of Al![Br_{3}[/tex} by consuming 2 moles of Al and 3 moles of [tex]Br_{2}](/tpl/images/0092/6201/f0357.png)
So, For 15.0 g of Al; we can calculate;
(15.0 g Al) X (1 mole Al / 26.98 g Al) X (3 moles / 2 moles Al) X (159.81 g / 1 mole )
= 133 g of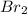 will be needed.
will be needed.
Therefore, 133g of will be needed to completely convert 15 g of Al into Al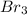 .
.