heartprague
06.05.2020 •
Chemistry
What is the molarity of a solution that contains 0.500 mole of kno3 dissolved in 0.500-liter of solution?
Solved
Show answers
More tips
- F Food and Cooking How to Make Shortcrust Pastry: Recipe and Tips...
- H Horoscopes, Magic, Divination Is there a 13th Zodiac Sign?...
- H Health and Medicine Want to Lose Weight? Gain Muscle without Damaging Your Health!...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...

Ответ:
Answer : The molarity of solution is, 1.00 M
Explanation : Given,
Moles of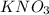 = 0.500 mol
= 0.500 mol
Volume of solution = 0.500 L
Molarity : It is defined as the number of moles of solute present in one liter of volume of solution.
Formula used :
Now put all the given values in this formula, we get:
Therefore, the molarity of solution is, 1.00 M
Ответ: