wusssup people or aliens im kinda bored ,, i already read my books on shelves but its still make me bored
Solved
Show answers
More tips
- L Legal consultation Juvenile Justice: Who Needs It?...
- F Family and Home How to Choose the Best Diapers for Your Baby?...
- F Family and Home Parquet or laminate, which is better?...
- L Leisure and Entertainment How to Properly Wind Fishing Line onto a Reel?...
- L Leisure and Entertainment How to Make a Paper Boat in Simple Steps...
- T Travel and tourism Maldives Adventures: What is the Best Season to Visit the Luxurious Beaches?...
- H Health and Medicine Kinesiology: What is it and How Does it Work?...
- O Other How to Choose the Best Answer to Your Question on The Grand Question ?...
- L Leisure and Entertainment History of International Women s Day: When Did the Celebration of March 8th Begin?...
- S Style and Beauty Intimate Haircut: The Reasons, Popularity, and Risks...
Answers on questions: History
- C Chemistry Which of the following is the affect an unconformity in rock layers has? A. It can cause continental drift. B. It can cause a volcanic eruption. C. It disrupts the geologic record....
- P Physics (a) calculate the total force of the atmosphere acting on the top of a table that measures 1.5 m 2.2 m. n (b) what is the total force acting upward on the underside of the table?...
- M Mathematics Set 1: 8, 15, 13, 7, 11, 17, 13 Median = 13 Lower quartile = 8 Upper quartile = 15 Range = 10 Interquartile range = 7 Set 2: 8, 15, 13, 7, 11, 17, 13, 100 Median = 13 Lower quartile...
- B Biology How many different populations live in the pond?...
- M Mathematics The cost of crawfish is $5.97 per pound. Which equation best represents y, the total cost of x pounds of crawfish?...

Ответ:
Join our video conference on Whereby
https://whereby.com/navneet175285
Ответ:
why aliens
Ответ:
Explanation:
Given parameters:
Mass of the sample = 0.622g
Volume of the gas = 2.4L
Temperature = 287K
Pressure of the gas = 0.85atm
Unknown
Molar mass of the gas = ?
Solution
To find the molar mass of the gas, we first assume that the gas in question is an ideal gas i.e. it obeys Boyle's and Charles's law.
On this premise, we can find the unknown which is the molar mass of the gas.
We first use the ideal gas equation to find the number of moles of the gas since all the parameters apart from the number of moles are given:Solving for number of moles:
The ideal gas equation is given as PV = nRT
We make the unknown "n" the subject of the equation
n =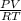
R is the gas constant whose value is 0.082atmdm³mol⁻¹K⁻¹
n =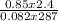
n =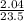 = 0.087mole
= 0.087mole
Now, we know the number of moles of the gas. We can proceed to find the molar mass from the given mass and the number of moles calculated using the equation below:molar mass =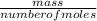
molar mass =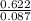 =7.15gmol⁻¹
=7.15gmol⁻¹
Part 2:
If the sample was placed under a low temperature, we want to consider what would happen to the volume:
Since we assume that the gas is an ideal gas, we can explain what will happen to the gas using Charles's law. The law states that "The volume of a fixed mass of a gas varies directly as its absolute temperature if the pressure is constant".
If we assume the pressure is constant for this gas, the volume of the gas would decrease as the temperature becomes lower. We would get lower values for both actual volume and predicted volume.